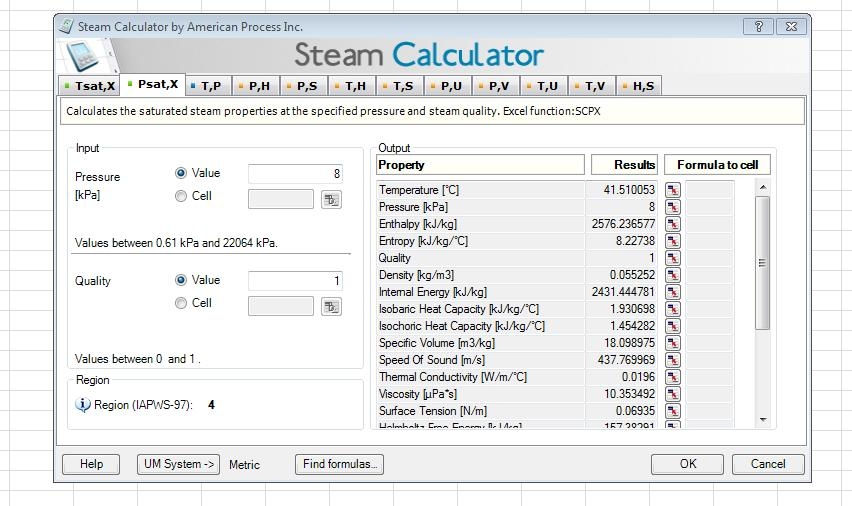
I believe it is free or reduced cost if you are an ASME member.
#Steam table thermodynamics calculator pdf
Second, to make full use of this you will want the ASME Steam Tables Compact Edition as a PDF that you can download from here: ASME Steam Tables, Compact Edition | eBooks Gateway | ASME Digital Collection. Λ = latent heat of vaporization of water at T oC i.e.First, the attached file is really a. Q = sensible heat supplied to unit weight of water to raise it from freezing point to to T oC expressed in Kcal/kg normal atmospheric pressure on the sea level at 0☌). (Atmospheric pressure at 1.01325 bar or kg/cm 2, i.e. Absolute pressure:Ībsolute pressure = Gauge pressure + Atmospheric pressure but it is very unstable and the steam soon resumes the saturated condition. This condition is obtained when it is cooled by its own expansion in a nozzle. The amount of superheating is measured by the rise in temperature of the steam above its saturation temperature. Superheating is assumed to take place at constant pressure. Steam in this condition, heated out of contact with water, is said to be superheated. The temperature of steam will then begin to increase with a corresponding increase in volume. If the water is entirely evaporated and further heat is then supplied, the first effect on the steam is to make it dry if it is not already dry. The steam having lesser temperature and greater density with respect to the steam table values for a particular saturation pressure is called super saturated steam. The quality of steam as regards its dryness is termed as dryness fraction. The steam in the steam space of a boiler generally contains water mixed with it in the form of a mist (fine water particles). is the reciprocal of specific volume.ĭry saturated steam is the sum of enthalpy of saturated water and enthalpy of evaporation and is defined as the quantity of heat required to raise the temperature of one kilogram of water from freezing point to the temperature of evaporation (corresponding to given pressure p)and then convert it into dry saturated steam at that temperature and pressure. Specific mass of the steam in a volume of one M 3. The value in cubic metre per kg of dry saturated steam (M 3/kg) Density of the steam: Total heat is the sum of the enthalpy of the various states, liquid (water) and gas (vapour) and also total heat can defined as the total amount of heat received by 1 kg of water from O oC at constant pressure to convert it to desired form of steam. It is the total heat contained in 1 kg of steam. Total Heat or Specific enthalpy of the steam: Sensible heat is related to changes in temperature of a gas or object with no change in phase. Latent heat is related to changes in phase between liquids, gases, and solids. Latent and sensible heat are types of energy released or absorbed in the atmosphere.
The heat that causes these changes is called latent heat. Solids can become liquids (ice to water) and liquids can become gases (water to vapor) but changes such as these require the addition or removal of heat. Latent heat (Enthalpy of Evaporation or vaporization)Īn amount of heat required to convert its phase of one kilogram of water at a given temperature.Īll pure substances in nature are able to change their state. Heat that causes a change in temperature in an object without phase change is called sensible heat. Similarly, when heat is removed from an object and its temperature falls, the heat removed is also called sensible heat. The increase in heat is called sensible heat. When an object is heated, its temperature rises as heat is added. Sensible heat is the energy required to change the temperature of a substance with no phase change. Generally it is expressed in Kcal/kg or KJ/kg Sensible Heat (Specific enthalpy of water): Enthalpy of evaporation is the difference between enthalpy of dry saturated steam and enthalpy of (saturated) water, i.e,Įnthalpy of evaporation = Enthalpy of dry saturated steam – Enthalpy of boiling water Similarly, the term total heat of superheated steam is now known as “enthalpy of superheated steam”. The terms formerly known as “ heat of water” (sensible heat), “ heat of evaporation” (latent heat) and “ total heat of steam” are now known as the “ enthalpy of (saturated) water”, “ enthalpy of evaporation” and “ enthalpy of saturated steam” respectively. (here not considered for use enthalpy in thermodynamics).
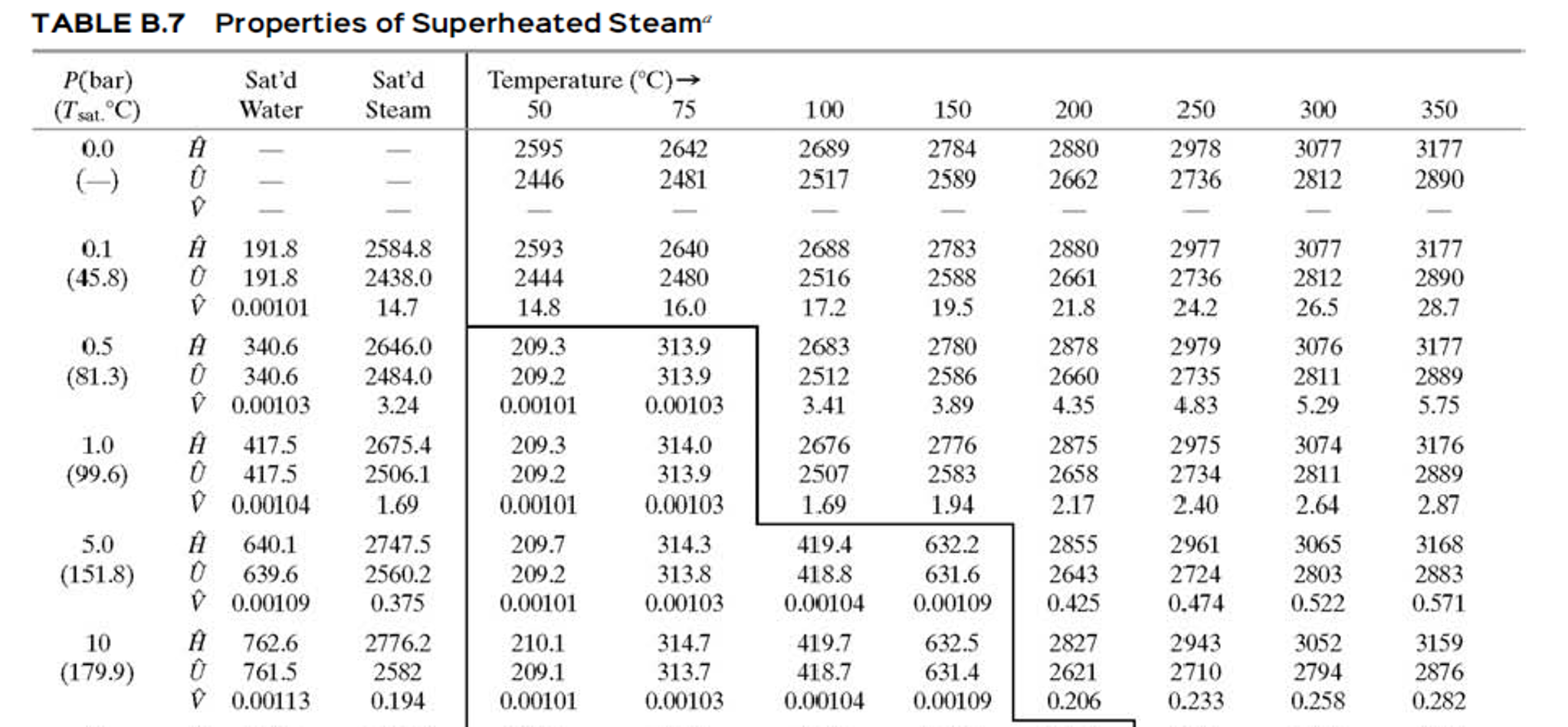
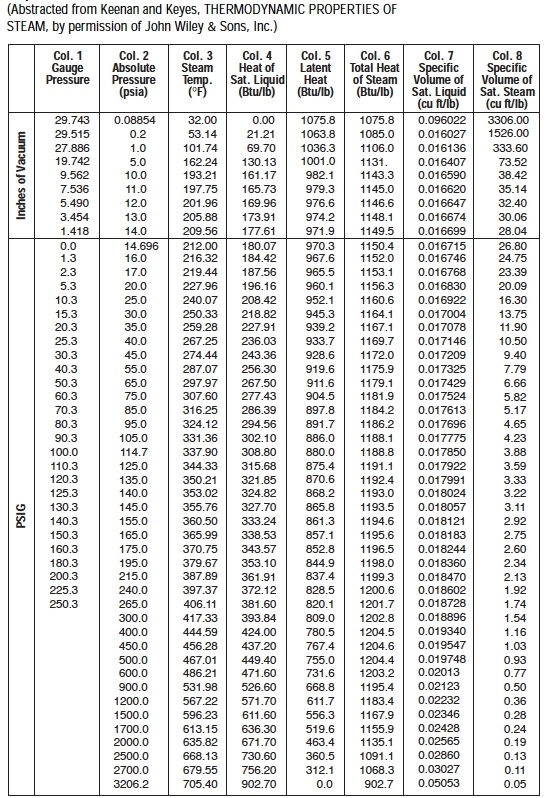
So we understand some definitions in steam properties:Įnthalpy can understand in the simple way by the following. In Sugar process industry Equipment Design and drawing calculations, the properties of saturated steam are involved at every step. 1.11.1 Online Calculator of Steam Table for Saturated Steam.1.4 Total Heat or Specific enthalpy of the steam:.1.3 Latent heat (Enthalpy of Evaporation or vaporization).1.2 Sensible Heat (Specific enthalpy of water):.1 Properties of Steam like Sensible Heat, Latent heat, Total Heat.

